|
Two new masters students have started in the lab this month. Rose Agnew is an MRes student using meta-analysis to investigate how climate affects fertility in wild birds, and Chris Carmichael is an MBiolSci student using meta-analysis to investigate the fitness benefits of sexual cannibalism in spiders and praying mantises. Welcome both!
0 Comments
Lily Amos has started her PhD in the lab in the last month. Lily will be investigating whether the strength of cryptic female choice in Drosophila melanogaster fruit flies is flexible in relation to a female's environment or health. Lily is co-supervised by Stu Wigby at Liverpool and Nicola Hemmings at Sheffield. Welcome Lily!
This week saw the publication of my latest meta-analysis project, which was in collaboration with Mike Skirrow, Mike Jennions and Leigh Simmons. We tested whether males exhibiting alternative 'sneaker' mating tactics show greater investment into sperm and ejaculate traits than males showing other mating tactics. The answer is: not really, but for interesting reasons! The paper is available from Biological Reviews here.
NEW! I am currently looking for a PhD candidate to join my lab at the University of Liverpool. I am offering a fully-funded project (3.5 years) examining how the environment influences female control of fertilisation in the fruit fly Drosophila melanogaster. The project is jointly supervised by me, Dr Stuart Wigby and Dr Nicola Hemmings.
The project is open to both UK and international students. Please see this page or contact me for more details. The deadline for applications is the 14th of January 2022. My new paper in Nature Ecology and Evolution has just been published online. This is the published version of the preprint I talked about recently. The take-home message is that males display to females less when they are in poor condition, and more when they are old. Therefore, sexual display behaviour generally reflects a male's condition, and so can be seen as 'honest'. This also suggests that females could often benefit from choosing males based on their display effort.
This week saw the publication of the first part of my fellowship project, in which I used meta-analysis to quantify the extent of context-dependent mating behaviour across the animal kingdom. I found that animals are significantly choosier during mate choice in favourable environments (with low predation rates, and a high mate density). However, both investment in animal sexual signalling behaviour, and the response to the sexual signals of others, were not dependent on the environment. Overall, this suggests that the evidence for context-dependent mating behaviour in the published literature is actually surprisingly weak. I discuss reasons why I think this is the case- but any opinions are welcome!
I played a small part in a recent paper published in Oikos, examining the current and potential future challenges facing teachers and students of ecology at all academic levels. This paper was the result of a fantastic meeting held at the Open University in 2019, bringing together a diverse group of teachers of ecology, and led by Julia Cooke and Zen Lewis. We identified a range of challenges facing students and teachers, including an increasing disconnect between society and nature, the expense and accessibility problems associated with running field trips, and the lack of diversity in ecology research. We also suggested solutions to these problems, many of which could be aided by new technologies.
I have recently uploaded two preprints to BioRxiv, from my fellowship project using meta-analysis to quantify the extent of context- and state-dependent mating behaviour in animals.
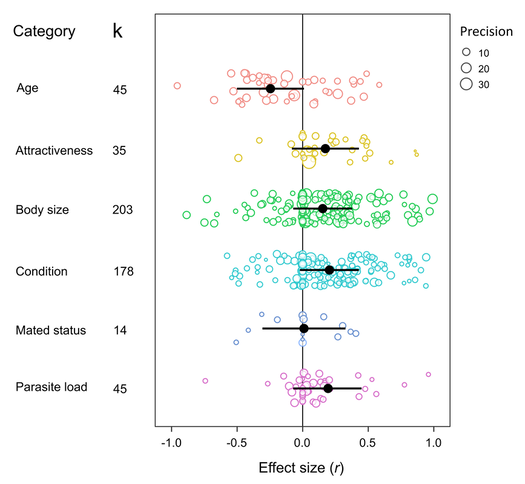
In the first, I present data from three meta-analyses (incorporating effec sizes from 222 studies in total) looking at how the expression of sexual signalling behaviour, response to sexual signals, and the strength of mate choice varies in relation to the environmental context. I find that mate choice is stronger when the environmental conditions are ‘friendly’ (e.g. no predators, lots of mates), but sexual signalling behaviour or responsiveness aren't affected by environmental conditions. Overall, this suggests that the current evidence for context-dependent mating behaviour across animals is actually surprisingly weak, and there is A LOT of variation across species and studies that is mostly unexplained. More work is needed to figure what is causing this variation. In the second preprint, I look at how individual state influences the expression of just sexual signalling behaviour (the choosiness and responsiveness data will be in a forthcoming paper). I show that, across 147 animal species, males signal more when they are attractive and in good condition. Therefore, signalling effort appears to be ‘honest’, and reflects the resources available to males. This means females could potentially use male signalling effort as an indicator of male quality. There was little evidence for terminal investment in signalling behaviour, except when looking at old virgins, who signalled more compared to young virgins. This week saw the publication of Alice Davies' masters project in Behavioral Ecology, in collaboration with me and Zen Lewis. In the paper we use meta-analysis to examine factors affecting the strength of mate-choice copying in animals. We found some evidence for taxonomic differences in copying strength, with mammals showing the strongest copying and invertebrates (primarily insects) showing the weakest copying. However, experimental design was amuch more important determinant of copying strength. Additionally, the strength of copying was not influenced by the sex of the copier, despite theory predicting otherwise. See the paper online and open-access here. Our analysis also overlaps with the recent meta-analysis by Jones & DuVal in Frontiers in Ecology and Evolution. So do check out that paper as well! This week saw the early-access publication of my new review where I discuss different experimental approaches to measuring mate choice. Mate choice is a tricky behaviour to measure, and I synthesise evidence showing how subtle differences in experimental design can have big impacts on how animals behaviour, and what we measure, during mate choice experiments. It's also my first single-author paper, and took a long time to write- the first draft was written back in 2015!
|
Archives
February 2024
AuthorLiam Dougherty. Categories |





 RSS Feed
RSS Feed
