|
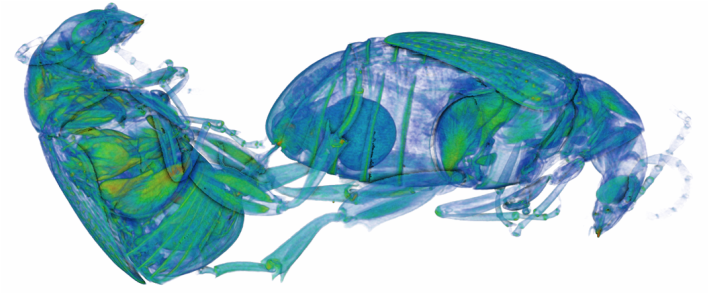
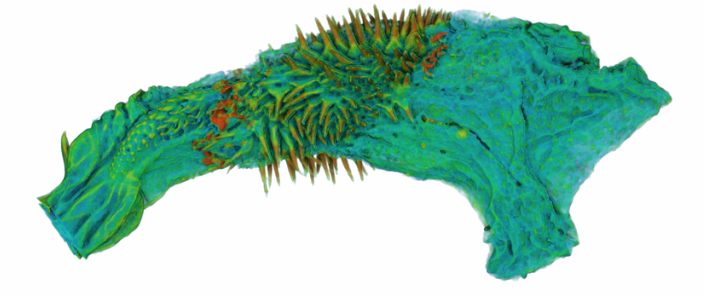
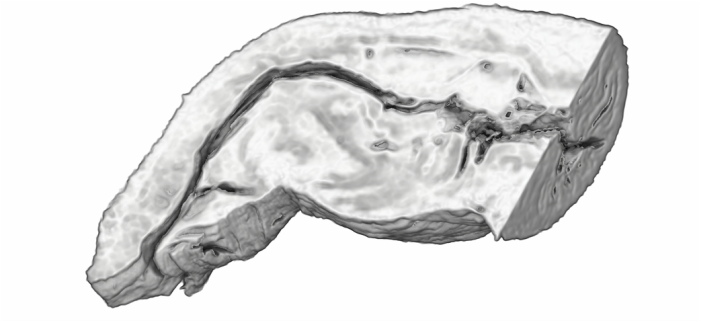
During the last year I have been working at UWA on the use of Micro-CT X-Ray scanning for studying the coevolution of insect genitalia. Micro-CT is not yet widely used in the field of genital evolution and we are hoping to be able to showcase the types of analysis that are possible when using modern scanning hardware and visualisation software. Micro-CT has several advantages over traditional anatomical techniques. First and foremost, it allows for the creation of three-dimensional datasets that can be shared online and viewed (and manipulated) by anyone using free open-source software. A key advantage for us is the ability to visualise internal anatomical structures without dissection, which runs the risk of damaging or altering the shape of such structures. In this post I would like to present a few examples of the kinds of visualisations that I have been doing, using as an example our work on the seed beetle Callosobruchus maculatus. Below is a visualisation of a pair of C. maculatus scanned during mating. The male is on the left and the female on the right. To create this image, a mating pair were first flash-frozen in liquid Nitrogen, so as to freeze the genitalia in place. The pair were then placed into a staining solution in order to increase the X-Ray contrast of the tissues, making analysis much easier. I have virtually removed the lowest density tissues, which has the result of making the cuticle transparent. The tissue has also been colour-coded by density, with highest density tissue in red and lowest in blue. The male leg muscles can be seen in red/yellow (muscle tissue is not very dense but takes up stain very well, resulting in high X-Ray absorbance). The large spherical structure in the female abdomen is the reproductive tract, which has expanded as the male has already deposited his spermatophore. C. maculatus males are probably best known for their penis, which is covered in sharp spines. Below is a visualisation of a C. maculatus penis that has been ‘virtually dissected’ from the female reproductive tract during mating. The rest of the male is to the left, and the head of the penis is to the right (sperm is released from here). Again this has been colour-coded by density. The spines can be seen in red, meaning they spines are made of a high density material when compared to the rest of the penis. Note also the number of spines; I counted around 230 on this male (it is hard to be exact because some spines are very small). During mating the spines on the penis stab into the wall of the female reproductive tract, leaving deep scars and wounds. This is harmful to the female, and can result in a significant reduction in female fecundity and even lifespan. Consequently, in this species the walls of the female reproductive tract are thickened when compared to closely related species in which the males lack penis spines. Below is a visualisation of the area of the female reproductive tract contacted by the penis spines during mating, which I have virtually sliced length-wise. The end of the female abdomen is to the left and the female head is to the right. The wall of the tract is very thick here when compared with the regions that are not in contact with the penis spines. The dark line in the centre is the lumen of the tract, which is very narrow when not mating (this is expanded greatly when the penis enters). I have been using micro-CT to measure changes in the thickness of the female reproductive tract in C. maculatus across populations and in response to different experimental conditions. Micro-CT has allowed us to do things previously not possible using dissection, such as calculating the volume of tissue in a given morphological trait. I have also been scanning pairs in copula to look at the interactions between male and female genitalia during mating, and the timing and location of female tract scarring. This is all still a work in progress but hopefully it will begin to be published soon, along with plenty of high resolution images and videos. For now watch this space for more info! NOTE: all of the above scans were performed using a ZEISS Versa 520 XRM at the University of Western Australia, and visualised using the AVIZO software package. All samples were stained with iodine prior to scanning. The voxel size is 7.4 μm3 for the mating pair, 1.45μm3 for the penis and 2.35 μm3 for the female tract.
2 Comments
|
Archives
February 2024
AuthorLiam Dougherty. Categories |
 RSS Feed
RSS Feed
